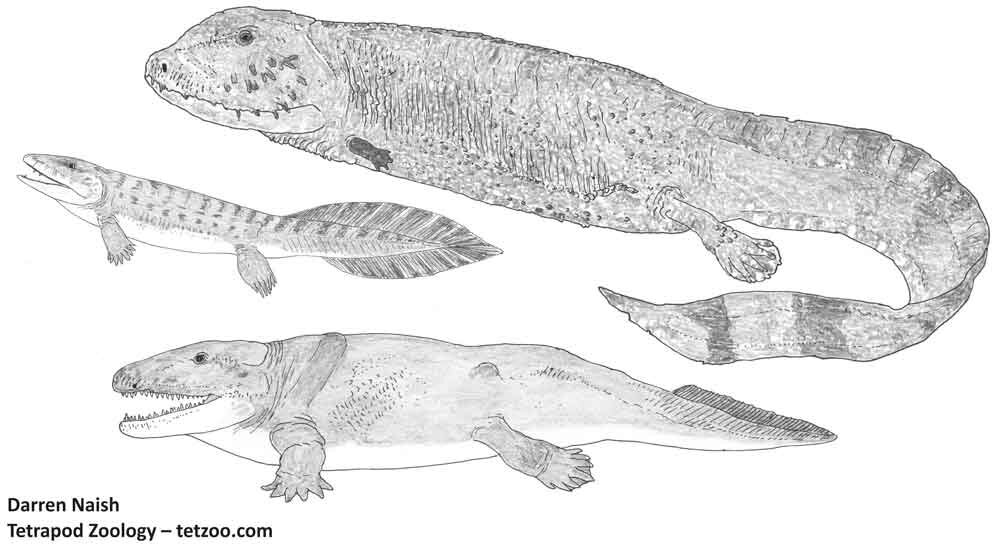
Long-time readers of Tetrapod Zoology will know of my long-term plans to complete a series of articles that cover the TOADS OF THE WORLD, the first part of which was published in 2009….
What, If Anything, Is A Strabomantid?
New Species Round-up for 2023, Part 1
This is a time of ecological crisis and massive loss of animal diversity, make no mistake about it. But there’s still a vast amount of new stuff left to discover, and every year we see a significant influx of newly recognized species, even among tetrapods. In this and the next article, we take a whistle-stop tour of those tetrapod species new to science as of 2023. As ever, remember that new to science is not synonymous with new to humanity…
The Amazing Caecilians
I have elected to republish my articles on caecilians. (1) Because TetZoo always needs more amphibian coverage. (2) Because caecilians are awesome. The text you’re about to read - assuming you choose to continue - first appeared in 2008 (original here); I haven’t much updated it. Here we go…
The Remarkably Weird Skeletons of Frogs
Tiny Frogs and Giant Spiders: Best of Friends
Allodapanura, the Biggest Frog Group You’ve Never Heard Of (Part 1)
A Love Letter to the Common Frog
Professor Jenny Clack, 1947-2020
Within recent decades, four specific areas of palaeontological discovery and reinterpretation have succeeding in capturing mainstream attention: the feathering of dinosaurs, the evolution of hominins, the early history of whales, and the early evolution, anatomy and biology of the earliest tetrapods. And it’s the last of those subjects we’re focusing on here. It’s a subject which has seen regular airing in top-tier journals, science magazines and TV documentaries, and one which has undergone a major and exciting revolution since the 1980s.
Caption: at left, Professor Jenny Clack, in the field at Burnmouth in the Scottish Borders. At right: in 2017, Jenny and her husband Rob got to fly in a Spitfire. Images: (c) Rob Clack, used with permission.
One person above all others has been responsible for leading research in this field and has made numerous ground-breaking discoveries herself, both in the field and laboratory. I am of course referring to Professor Jenny Clack of the Department of Zoology at the University of Cambridge, an excellent and highly respected scientist whose technical papers are authoritative, ground-breaking and of the highest standard. Her publication list is formidable. Alas, I regret to write that I’m here for sad reasons, since Jenny died on the morning of 26th March 2020 following a five-year struggle with cancer. While – for my shame – I haven’t written about early tetrapod evolution here at TetZoo before, nor about Jenny’s research specifically, both are areas I’ve avidly followed, and I want to share some brief thoughts here.
Caption: a selection of Clack publications in the TetZoo library. Image: Darren Naish.
Prior to Jenny’s work, the consensus view on the oldest tetrapods – in particular Ichthyostega from the Devonian of Greenland – is that these were ‘terrestrialised’, vaguely salamander-like quadrupeds, well able to walk on land by planting all four feet on the substrate. This view, entrenched in textbooks and the popular literature, mostly owed itself to the work of Swedish palaeontologist Erik Jarvik whose work on Ichthyostega, while initiated in the 1950s, took some decades to appear, finally being published in 1996. Jarvik’s work has its own long and curious backstory (Ries 2007) – which I can’t cover here – and it certainly wasn’t missed by some of us that the conclusions of his supposedly definitive monograph were being called into doubt just weeks after its appearance (Norman 1996).
Caption: Jenny Clack (and Professor Robert Insall) at the Ballagan Formation type locality, near Glasgow. Image: (c) Rob Clack, used with permission.
Tantalising remains of an Ichthyostega contemporary – the smaller Acanthostega – were discovered in East Greenland on a Cambridge University expedition of 1970 (and were languishing, unrecognised, in the drawers of the university’s earth sciences department prior to Jenny’s interest). These indicated that, with luck, more early tetrapod finds might be discovered in the same region. After making special arrangements with Danish scientists – and with Jarvik, who the Danes wanted to remain on good terms with – Jenny visited Greenland in 1987; she was accompanied by colleagues from Copenhagen, her husband Rob and her then PhD student Per Ahlberg (Clack 1988). They were remarkably successful, retrieving multiple good specimens, and continued to be so on later trips to the same region.
Caption: in more recent years, it’s become obvious that Ichthyostega - the classic ‘early tetrapod’ - was not just a formidable, toothy predator, but an unusual, specialised animal with paddle-like hindlimbs, a proportionally short tail and a regionalised vertebral column. Image: Ahlberg et al. (2005).
Was Jarvik right about these animals? Not really, no. The discovery of numerous aquatic specialisations in Ichthyostega and the smaller Acanthostega – published by Mike Coates and Jenny during the early 1990s (Coates & Clack 1990, 1991) – was a huge surprise. Tetrapods didn’t start their history as land-walking animals with pentadactyl hands and feet, as Jarvik (and just about everyone else) had thought, but were aquatic and polydactyl, with 8 fingers and 7 toes (or thereabouts)! Later work showed that Ichthyostega had an unusual ear perhaps specialised for aquatic hearing (Clack et al. 2003) and a remarkable regionalised vertebral column and other peculiarities (Ahlberg et al. 2005) suggesting that, when on land, it might have moved in mudskipper-like fashion (Ahlberg et al. 2005, Pierce et al. 2012, 2013).
Caption: life reconstructions of three early tetrapods worked on by Clack: the big Crassigyrinus, the small, aquatic Acanthostega, and the big (c 1 m long) Ichthyostega. These images are among tens of archaic tetrapods reconstructed for my in-prep textbook. I’ve learnt recently that the Crassigyrinus will soon need revising. Image: Darren Naish.
Jenny’s work wasn’t limited to this fundamental reinterpretation of the earliest tetrapods, but also involved taxa from throughout the Devonian and Carboniferous. She revised knowledge of the remarkable aquatic Carboniferous tetrapod Crassigyrinus (Clack 1998a), described the new species Eucritta melanolimnetes (Clack 1998b), Pederpes finneyae (Clack 2002a, Clack & Finney 2005), Kyrinion martilli (Clack 2003) and others, reported entire new faunal assemblages (Clack et al. 2016) and published key interpretations of such groups as embolomeres (Clack 1987, Clack & Holmes 1988), chroniosuchians (Clack & Klembara 2009, Klembara et al. 2010) and microsaurs (Clack 2011). This extensive, substantial experience made her the right person to publish an authoritative and comprehensive book on Devonian and Carboniferous tetrapods: Gaining Ground: The Origin and Evolution of Tetrapods appeared in 2002 (Clack 2002). It went to second edition in 2012. It’s the best guide to early tetrapod evolution and fills a much-needed gap in the semi-technical literature, and I strongly recommend it to those interested in the subject.
Caption: covers of the first and second editions of Clack’s Gaining Ground. The cover images are, respectively, by Jenny Clack and Julia Molnar. The Clack image shows two individuals engaging in courtship: the green hump at the back left is the body of a second animal.
I never had the privilege of working with Jenny or of accompanying her in the field, but I did have reason to speak to her on several occasions, and to correspond with her. She was friendly and generous with her time and went to trouble to provide me with information and illustrations on an occasion when email wasn’t doing its job. It’s also obvious that she had a sense of humour. Of those new species listed above, Eucritta melanolimnetes translates roughly as ‘creature from the black lagoon’. Some – probably all – of the Ichthyostega and Acanthostega specimens she discovered in Greenland had nicknames: I’m pretty sure I recall seeing that one of them was called Grace Jones. Those who knew her so much better confirm that she was excellent fun, a great leader in the field, and a brilliant mentor.
Caption: several of the archaic Devonian tetrapods study by Clack and her colleagues are excellent, 3D and with very detailed preservation. This image shows an Acanthostega gunnari cast at Musee De L'Histoire Naturelle, Brussels. Image: Ghedoghedo. CC BY-SA 3.0 (original here).
Jenny began her scientific career during the early 1970s and received her PhD at the University of Newcastle Upon Tyne in 1984 under Alec Panchen. Panchen initially offered her a PhD following anatomical discoveries she made while preparing the notoriously difficult holotype specimen of the Carboniferous embolomere Pholiderpeton, initially described by Thomas Huxley in 1869. She joined the University of Cambridge in 1981 and for more than ten years was Assistant Curator at the University’s Museum of Zoology, later being promoted to Senior Assistant Curator and then Curator. She supervised a number of people who went on to become well known in vertebrate palaeontology and evolutionary biology, among them Mike Lee (whose PhD was specifically on pareiasaurs), sauropod expert Paul Upchurch, and actinopterygian fish worker Matt Friedman.
Caption: life-sized models of Ichthyostega and Acanthostega have been made a few times. Here’s the Acanthostega on show at the Sedgwick Museum, Cambridge. I’ve photographed this model several times but none of my photos are that good - I stole this image from Christian Kammerer (source).
Jenny retired in 2015. As you might expect give her scientific achievements, she was highly decorated, holding several palaeontological medals, additional, honorary doctorates and other awards too. Such was mainstream interest in her work and ideas that a 2012 episode of the BBC series Beautiful Minds was devoted to her (it’s viewable here on youtube); among other interesting things, it reveals that she was inspired during her childhood years by newts and freshwater fishes, and that it was learning about Mary Anning which encouraged her to pursue palaeontology. She liked motorbikes and cats, and some of the documentaries that focus on her work show her riding around on her motorbike.
Caption: the Tournaisian rocks of Burnmouth, north of Berwick-upon-Tweed, have, within recent decades, proved an important new locality for tetrapods. As a consequence, Jenny and colleagues set up the TW:eed Project, the acronym standing for Tetrapod World: Early Evolution and Diversity. Jenny (in the green top) stands close to the middle. Image: (c) Rob Clack, used with permission.
My text here only touches on a few aspects of her achievements, life and technical contributions, and I know that much else could be said. I’m very saddened to hear of her passing, very much regarded her as an excellent scientist who published exciting research, and I’ll miss her as a regular attendee of palaeontological meetings. My sincere condolences to her husband Rob, and to those others who knew and loved her.
Some of the text here is adapted from my in-prep giant textbook on the vertebrate fossil record.
The University of Cambridge website on Professor Clack is here
An excellent website on Professor Clack’s research and expeditions is here
Refs - -
Ahlberg, P. E., Clack, J. A. & Blom, H. 2005. The axial skeleton of the Devonian tetrapod Ichthyostega. Nature 437, 137-140.
Clack, J. A. 1987. Two new specimens of Anthracosaurus (Amphibia: Anthracosauria) from the Northumberland coal measure. Palaeontology 30, 15-26.
Clack, J. A. 1988. Pioneers of the land in East Greenland. Geology Today 4 (6), 192-194.
Clack, J. A. 1998a. The Scottish Carboniferous tetrapod Crassigyrinus scoticus (Lydekker) – cranial anatomy and relationships. Transactions of the Royal Society of Edinburgh: Earth Sciences 88, 127-142.
Clack, J. A. 1998b. A new Early Carboniferous tetrapod with a mélange of crown-group characters. Nature 394, 66-69.
Clack, J. A. 2002a. An early tetrapod from 'Romer's Gap'. Nature 418, 72-76.
Clack, J. A. 2003. A new baphetid (stem tetrapod) from the Upper Carbinoferous of Tyne and Wear, U.K., and the evolution of the tetrapod occiput. Canadian Journal of Earth Sciences 40, 483-498.
Clack, J. A. 2011. A new microsaur from the Early Carboniferous (Viséan) of East Kirkton, Scotland, showing soft tissue evidence. Special Papers in Palaeontology 86, 45-55.
Clack, J. A., Ahlberg, P. E., Finney, S. M., Dominguez Alonso, P., Robinson, J. & Ketcham, R. A. 2003. A uniquely specialized ear in a very early tetrapod. Nature 425, 65-69.
Clack, J. A., Bennett, C. E., Carpenter, D. K., Davies, S. J., Fraser, N. C., Kearsey, T. I., Marshall, J. E. A., Millward, D., Otoo, B. K. A., Reeves, E. J., Ross, A. J., Ruta, M., Smithson, K. Z., Smithson, T. R. & Walsh, S. A. 2016. Phylogenetic and environmental context of a Tournaisian tetrapod fauna. Nature Ecology & Evolution 1: 0002.
Clack, J. A. & Finney, S. M. 2005. Pederpes finneyae, an articulated tetrapod from the Tournaisian of western Scotland. Journal of Systematic Palaeontology 2, 311-346.
Clack, J. A. & Holmes, R. 1988. The braincase of the anthracosaur Archeria crassidisca with comments on the interrelationships of primitive tetrapods. Palaeontology 31, 85-107.
Clack, J. A. & Klembara, J. 2009. An articulated specimen of Chroniosaurus dongusensis and the morphology and relationships of the chroniosuchids. Special Papers in Palaeontology 81, 15-42.
Coates, M. I. & Clack, J. A. 1990. Polydactyly in the earliest known tetrapod limbs. Nature 347, 66-69.
Coates, M. I. & Clack, J. A. 1991. Fish-like gills and breathing in the earliest known tetrapod. Nature 352, 234-236.
Klembara, J., Clack, J. A. & Čerňanský, A. 2010. The anatomy of palate of Chroniosuchus dongusensis (Chroniosuchia, Chroniosuchidae) from the Upper Permian of Russia. Palaeontology 53, 1147-1153.
Norman, D. 1996. [Review of] The Devonian tetrapod Ichthyostega. Palaeontological Newsletter 31, 13-15.
Pierce, S. E., Ahlberg, P. E., Hutchinson, J. R., Molnar, J. L., Sanchez, S., Tafforeau, P. & Clack, J. A. 2013. Vertebral architecture in the earliest stem tetrapods. Nature 494, 226-229.
Pierce, S. E., Clack, J. A. & Hutchinson, J. R. 2012. Three-dimensional limb joint mobility in the early tetrapod Ichthyostega. Nature 486, 523-526.
Ries, C. J. 2007. Inventing ‘the four-legged fish’. The palaeontology, politics and popular interest of the Devonian tetrapod Ichthyostega, 1931-1955. Ideas in History 2, 37-78.
The Most Amazing TetZoo-Themed Discoveries of 2018
As we hurtle toward the end of the year – always a scary thing because you realise how much you didn’t get done in the year that’s passed – it’s time to look back at just a little of what happened in 2018...
Caption: animals we will meet below, a montage. Images: (c) Philippe Verbelen, (c) Kristen Grace, Florida Museum of Natural History, Graham et al. (2018), CC BY-SA 4.0.
This article is not anything like a TetZoo review of 2018 (I’ll aim to produce something along those lines in early 2019), but, rather, a quick look at some of the year’s neatest and most exciting zoological (well, tetrapodological) discoveries. As per usual, I intended to write a whole lot more – there are so many things worthy of coverage – and what we have here is very much an abridged version of what I planned.
Thanks as always to those supporting me at patreon. Time is the great constraint (and finance, of course), and the more support I have, the more time I can spend on producing blog content. Anyway, to business…
The Rote leaf warbler. New passerine bird species are still discovered on a fairly regular basis; in fact three were named in 2018*. One of these is especially remarkable. It’s a leaf warbler, or phylloscopid, endemic to Rote in the Lesser Sundas, and like most members of the group is a canopy-dwelling, insectivorous, greenish bird that gleans for prey among foliage. Leaf warblers are generally samey in profile and bill shape, so the big deal about the new Rote species – the Rote leaf warbler Phylloscopus rotiensis – is that its bill is proportionally long and curved, giving it a unique look within the group. It superficially recalls a tailorbird. Indeed, I think it’s likely that the species would be considered ‘distinct enough’ for its own genus if there weren’t compelling molecular data that nests it deeply within Phylloscopus (Ng et al. 2018).
* The others are the Cordillera Azul antbird Myrmoderus eowilsoni and the Western square-tailed drongo Dicrurus occidentalis.
Caption: a Common chiffchaff Phylloscopus collybita encountered in western Europe, a familiar Eurasian-African phylloscopid leaf warbler. Image: Darren Naish.
The story of the Rote leaf warbler’s discovery is interesting in that it’s yet another recently discovered species whose existence and novelty was suspected for a while. Colin Trainor reported leaf warblers on Rote in 2004 but never got a good look at them, Philippe Verbelen observed them in 2009 and realised how anatomically unusual they were, and it wasn’t until 2015 that a holotype specimen was procured (Ng et al. 2018). I’ve mentioned before the fact that documenting and eventually publishing a new species is rarely an instant see it catch it publish it event, but a drawn-out one that can take decades, and here we are again. Also worth noting is that the existence of a leaf warbler on Rote was not predicted based on our prior knowledge of leaf warbler distribution in view of the deep marine channel separating Rote from Timor and lack of any prior terrestrial connection. Yeah, birds can fly, but members of many groups prefer not to cross deep water channels. In this case, this did, however, happen and most likely at some point late in the Pliocene (Ng et al. 2018).
Caption: Rote leaf warbler in life, a novel member of an otherwise conservative group. Image: (c) Philippe Verbelen.
Rote has yielded other new passerines in recent years – the Rote myzomela Myzomela irianawidodoae (a honeyeater) was named in 2017 – and it’s possible that one or two others might still await discovery there.
Neanderthal cave art. Hominins don’t get covered much at TetZoo, which is weird given the amazing pace of relevant recent discoveries and the fact that they’re totally part of the remit. I mostly don’t cover them because I feel they’re sufficiently written about elsewhere in the science blogging universe, plus I tend to be preoccupied with other things. Nevertheless, I take notice, and of the many very interesting things published in 2018 was Hoffman et al.’s (2018) announcement of several different pieces of Spanish rock art, seemingly made by Neanderthals Homo neanderthalensis. The art concerned involves hand stencils, abstract lines, squares and amorphous patches of pigment, always marked in red.
Caption: red abstract markings, discovered in several Spanish caves, are old, and in fact were seemingly made by hominins long before H. sapiens moved into Europe. The red sinuous marking and system of squares and lines near the middle of this photo are purported to have been made by Neanderthals (other images, depicting animals and present adjacent to these markings, were seemingly created more recently by H. sapiens individuals). Image: (c) P. Saura.
The main reason for the attribution of this art to Neanderthals is its age: uranium-thorium dating shows that it’s older than 64ka, which therefore makes it more than 20ka older than the time at which H. sapiens arrived in Europe (Hoffman et al. 2018). That seems compelling, and it’s consistent with a building quantity of evidence for Neanderthal cultural complexity which involves the use of shells, pigments, broken stalagmites and so on.
Caption: one of the most famous pieces of claimed Neanderthal rock art: the Gorham's Cave ‘hashtag’ from Gibraltar. Image: (c) Stewart Finlayson.
I should add here, however, that I’m slightly sceptical of the use of age as a guide to species-level identification. Why? Well, we have evidence from elsewhere in the fossil record that the range of a hominin species can be extended by around 100ka without serious issue (witness the 2017 announcement of H. sapiens remains from north Africa; a discovery which substantially increased the longevity of our species). In view of this, would a 20ka extension of H. sapiens’ presence in Europe be absolutely out of the question? Such a possibility is not supported by evidence yet, and I don’t mean to appear at all biased against Neanderthals.
A tiny Cretaceous anguimorph in amber, and other Mesozoic amber animals. As you’ll know if you follow fossil-themed news, recent years have seen the discovery of an impressive number of vertebrate fossils in Cretaceous amber, virtually all of which are from Myanmar and date to around 99 million years old. They include tiny enantiornithine birds, various feathers (most recently racquet-like ‘rachis dominated feathers’), the tiny snake Xiaophis, early members of the gecko and chameleon lineages and the small frog Electrorana. Many of these finds were published in 2018 and any one could count as an ‘amazing’ discovery.
Caption: the Barlochersaurus winhtini holotype, from Daza et al. (2018).
However, there’s one fossil in particular that I find ‘amazing’, and it hasn’t received all that much coverage. It’s the tiny (SVL* 19.1 mm!), slim-bodied anguimorph Barlochersaurus winhtini, named for a single, near-complete specimen subjected to CT-scanning (Daza et al. 2018). Remarkable images of its anatomical details are included in Daza et al.’s (2018) paper. It has short limbs, pentadactyl hands and feet and a slim, shallow, bullet-shaped skull. Phylogenetic study finds it to be somewhere close to, or within, Platynota (the clade that includes gila monsters and kin, and monitors and kin), or perhaps a shinisaurian (Daza et al. 2018). It could be a specialised dwarf form, or somehow more reflective of the ancestral bauplan for these anguimorph groups. Either way, it’s exciting and interesting. What next from Burmese amber?
* snout to vent length
Caption: Barlochersaurus in life. It’s about the size of a paperclip. Image: (c) Kristen Grace, Florida Museum of Natural History (original here).
The Reticulated Siren. Sirens are very special, long-bodied aquatic salamanders with reduced limbs and bushy external gills. They’re very weird. They can reach 95 cm in length (and some fossil species were even larger), lack hindlimbs and a pelvis, have a horny beak and pavements of crushing teeth, and eat plants in addition to gastropods, bivalves and other animal prey. A longish article on siren biology and evolution can be found here at TetZoo ver 3.
Caption: a life reconstruction of the Cretaceous siren Habrosaurus, showing features typical of the group. This animal could reach 1.5 m in total length. Image: Darren Naish (prepared for my in-prep texbook The Vertebrate Fossil Record, on which go here).
Until recently, just four living siren species were recognised. But it turns out that indications of a fifth – endemic to southern Alabama and the Florida panhandle – have been around since 1970 at least. Furthermore, they pertain to a big species, similar in size to the Great siren Siren lacertina. Known locally as the ‘leopard eel’ (a less than ideal moniker, given that there’s a real eel that already goes by this name), this animal has been published by Sean Graham and colleagues in the open-access journal PLoS ONE (Graham et al. 2018) wherein it’s formally christened the Reticulated siren S. reticulata. It reaches 60 cm in total length, has dark spots across its dorsal surface and a proportionally smaller head and longer tail than other Siren species.
A museum specimen of the species has been known since 1970 when its finder noted that it did “not conform” to descriptions of known species, and live specimens were collected by David Steen and colleagues in 2009 and 2014. Again, note that discovery and recognition was a drawn-out process. The discovery has, quite rightly, received a substantial amount of media coverage, and many interesting articles about the find are already online. Many of you will already know of David Steen due to his social media presence and Alongside Wild charity (which I’m proud to say I support via pledges at patreon).
Caption: the Reticulated siren paratype specimen, as described by Graham et al. (2018). Image: Graham et al. (2018), CC BY-SA 4.0. Original here.
The idea that a new living amphibian species 60 cm long might be discovered anew in North America in 2018 is pretty radical. I’m reminded of the 2009 TetZoo ver 2 article ‘The USA is still yielding lots of new extant tetrapod species’ (which is less fun to look at than it should be, since images aren’t currently showing at ver 2). Furthermore, Graham et al. (2018) discovered during their molecular phylogenetic work that some other siren species are not monophyletic but likely species complexes, in which case taxonomic revision is required and more new species will probably be named down the line.
And that’s where I must end things, even though there are easily another ten discoveries I’d like to write about. This is very likely the last article I’ll have time to deal with before Christmas. As I write, I’m preparing to leave for the Popularising Palaeontology conference which happens in London this week (more info here), and then there are Christmas parties and a ton of consultancy jobs to get done before the New Year. On that note, I’ll sign off with a festive message, as is tradition. Best wishes for the season, and here’s to a fruitful and action-packed 2019. Special thanks once again to those helping me out at patreon.
For previous TetZoo articles relevant to various of the subjects covered here, see…
The USA is still yielding lots of new extant tetrapod species, July 2009
THE AMAZING WORLD OF SALAMANDERS, October 2013
Chiffchaffs: a view of passerines from the peripheries (part I), August 2014
The Biology of Sirens, June 2016
Refs - -
Daza, J. D., Bauer, A. M., Stanley, E. L., Bolet, A., Dickson, B. & Losos, J. B. 2018. An enigmatic miniaturized and attenuate whole lizard from the mid-Cretaceous amber of Myanmar. Breviora 563, 1-18.
Hoffman, D. L., Standish, C. D., García-Diez, M., Pettitt, P. B., Milton, J. A., Zilhão, J., Alcolea-González, J. J., Cantelejo-Duarte, P., Collado, H., de Balbín, R., Lorblanchet, M., Ramos-Muñoz, J., Weniger, G.-Ch. & Pike, A. W. G. 2018. U-Th dating of carbonate crusts reveals Neandertal origin of Iberian cave art. Science 359, 912-915.
New Living Animals We Want to Find
As a regular denizen of the TetZooniverse, you may well remember the July 2017 article ‘Fossils We Want to Find’ in which I discussed a list of hypothetical fossil things that we might one day discover but haven’t yet. Wouldn’t it be fun to do the same sort of thing with extant species; that is, with discoveries pertaining to living, breathing animals? Over at the Zoology for Enthusiasts facebook group (a spinoff of the Tetrapod Zoology facebook group), Jordan Fryer suggested doing exactly this, and consequently people have been coming up with their own suggested living animals that might await discovery. Because this seemed like a lot of fun (and a chance to discuss some really neat and unusual stuff), I thought I’d give it a go.
Caption: the 2017 precursor to the article you’re reading here was all about fossil animals. It included this photo, which shows me in the act of discovering a dinosaur bone in the Moroccan Sahara. Image: Richard Hing.
Naturally, any list of this sort is horribly subjective, reflecting the interests and biases of the person compiling the list, but so be it. It also seems all too easy to turn any compilation into a ‘list of most discoverable cryptids’: for those of you who don’t know, I have a long-standing interest in cryptozoology and have published on it quite frequently (see Naish (2017) for starters). For the most part, I’ve not done this, though read on.
Caption: many of my thoughts on mystery animals can be found in my 2017 book Hunting Monsters. I am not - sorry - much impressed by the case for such supposed animals as the mokele-mbembe, an artistic reconstruction of which is shown at right. Image: David Miller, in Mackal (1987).
I’ve also mostly excluded hypothetical discoveries that are inspired by the creatures of cryptozoology but could arguably be considered independent of the cryptozoological literature. In part this is because I don’t think they’re plausible or worth considering, but it’s also because they’re cliched and the opposite of original. So, no ‘living sauropods from the Congo’ or ‘living plesiosaurs in Loch Ness’, for example.
As for what I have selected: well, some of my suggestions are sillier than others, and some are perhaps not that interesting to non-specialists. But, whatever. Feel free to dissect my suggestions in the comments, and perhaps come up with your own.
Caption: among my suggested ‘fossils we want to find’ are protobats (like the hypothetical examples shown at left, from Graham (2002)) and a good skeleton of the giant hominid Gigantopithecus blacki. This ilustration of a lower jaw is from Simons & Ettel's (1970) magazine article. Images: Graham (2002), Simons & Ettel (1970).
A habitually bipedal, large, non-human hominid. Whatever you think of all those stories, anecdotes and sightings about bigfoot, yeti, almas, orang-pendek, yowie and so on and on, the fact remains that the discovery of a large, bipedal non-human hominid – whether it be a pongine, hominine, or member of another hominid lineage – would be a huge deal. It would not just be one of the most newsworthy creatures to ever be discovered; it would also have enormous ramifications for our understanding of hominid evolution and potentially the human condition itself.
Caption: are crypto-hominids a cultural phenomenon more than a zoological one? I’ve argued for both possibilities at different times. Whatever… for the purposes of the article you’re reading now, I hope we can agree that the discovery of such an animal would be high on any hypothetical ‘wants’ list. Image: Darren Naish.
It would also – if relating to North America or northern Eurasia in particular – very likely have a significant impact on economy, land management and land use in those regions… or, you’d hope it would, anyway (who knows, given the current state of environmental protection in the USA). The hypothetical discovery of such an animal would also be regarded by many as one of the biggest ‘wins’ ever scored against ‘establishment science’, and thus could well be a bad thing (viz, “if scientists were wrong about this, what else could they be wrong about?”). And I’ll stop there before we dive into a rabbit-hole of conspiracy theories and coverups.
A big, flightless passerine. The majority of living bird species – over 60% of them – are passerines, or perching birds. This is the great group that includes crows, thrushes, warblers, finches, sparrows and so many others. For all their success, wide distribution and diversity, passerines are generally quite samey. There are no big, long-legged wading passerines, or heavy-bodied diving passerines or flightless running passerines, for example. Why this is so remains mysterious: passerines didn’t take to those niches because… well, they just didn’t. Does this mean that they couldn’t? As usual, we can come up with a few reasons as to why they were ‘constrained’ in evolutionary potential, but any one of those reasons could be overturned by some evolutionary deviant that refuses to pay attention to the rules.
Caption: passerine birds are diverse, to a degree… here’s just a sample of their diversity. This is part of a giant montage that’s being built for my in-prep textbook The Vertebrate Fossil Record. Image: Darren Naish.
And thus I submit that a particularly large, wholly flightless, cursorial passerine should make itself known to the world. It should be a record-holder as goes size, but not necessarily be that much bigger than the largest known passerines (like lyrebirds and ravens): I’m talking about a bird that weighs 3-5 kg and is thus similar in size to a large chicken. It should be a big, long-legged rail-babbler, quail-thrush or similar, and hence be a denizen of Wallacea or nearby.
Caption: Eupetes, the Malaysian rail-babbler. A hypothetical big, flightless passerine should be a close relative of this bird. Image: Francesco Verronesi, CC BY-SA 2.0 (original here).
A few recently extinct, island-dwelling passerines were flightless, so we do know that passerines have the evolutionary potential to follow this pathway. Such species (a bunting and a few New Zealand wrens… and possibly a few others) were all small (less than 40 g).
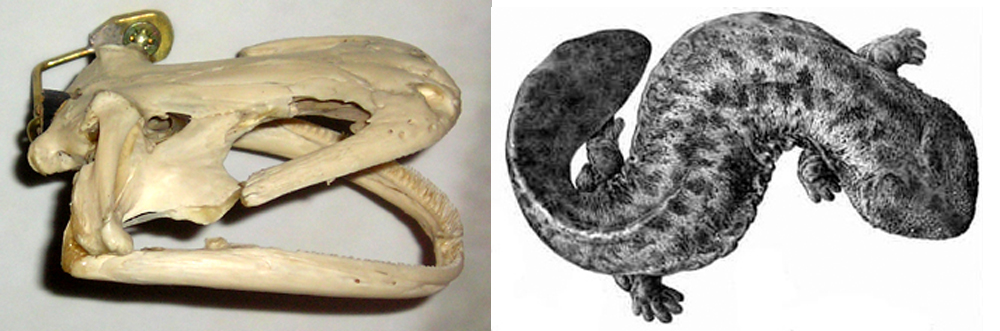
A western Asian giant salamander. Giant salamanders (cryptobranchids) are restricted today to eastern Asia (where Andrias occurs) and North America (where Cryptobranchus occurs). Hunting, human disturbance, habitat loss and deterioration, climate change and other issues are putting them into perilous decline, right at the same time as we’re discovering that some of them are species complexes. They were more widespread in the past than they are today, since fossils show that Andrias salamanders were widespread across Europe and Asia between about 28 and 2 million years ago.
Caption: an Asian giant salamander (Andrias) photographed in captivity. Record-holding specimens of Andrias can be 1.8 m long and exceed 60 kg, and some extinct species reached even larger sizes. Image: Markus Bühler.
While there are very good reasons for the decline and extinction of the animals in the areas concerned, some of the regions where they formerly occurred still have what look like suitable habitat today and are sparsely populated by people. Furthermore, extinct giant salamanders weren’t all denizens of fast-flowing, highly oxygenated streams like those inhabited by the modern populations. Some inhabited ponds and lakes. Ergo: I would really, really like there to be a west Asian cryptobranchid that comes from a habitat considered weird for the other living members of the group. And it doesn’t have to be a giant of 2 metres or more. A hellbender-sized species of 70 cm or so will do fine thank you very much.
Caption: some extinct cryptobranchids - this is Zdeněk Burian’s reconstruction of Andrias scheuchzeri - inhabited European ponds and lakes. I’ve previously criticised this image for showing the animal as terrestrial. Since then, the proposal has been made that some extinct cryptobranchids (albeit not A. scheuchzeri) were significantly more terrestrial than living species. Image: (c) Zdeněk Burian.
Again, this is an area of special interest to cryptozoologists, since there have been occasional suggestions that stories, engravings and such from western Asia might reflect folk knowledge of unusually big salamanders in the region. In reality, the images and stories concerned are super-ambiguous and more likely refer to otters and god knows what else.
Caption: at left, an Andrias skull. Image: Darren Naish. At right: Japanese giant salamander (A. japonicus) illustration by Y. de Hoev from 1887. Image: Y. de Hoev, public domain (original here).
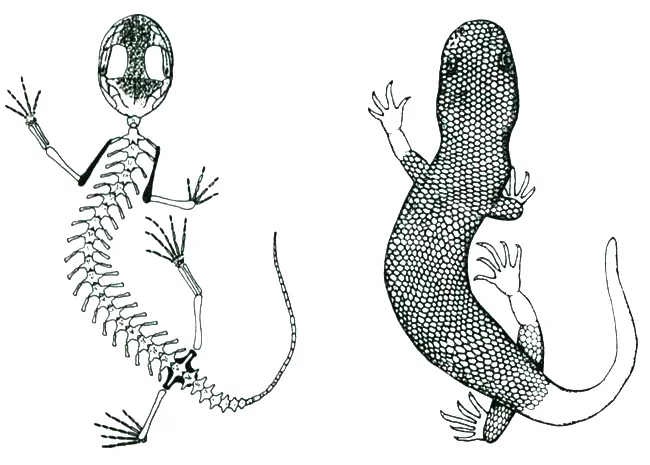
A living albanerpetontid. Everyone knows that there are three main groups of living amphibian: caecilians, salamanders and anurans (frogs and toads). But until (geologically) recently, there was a fourth group: the albanerpetontids, sometimes termed albies by those who work on them. Albanerpetontids were geographically widespread, their range including Eurasia, northern Africa and North America, and they were geologically long-lived too. The oldest are from the Middle Jurassic while the youngest are… well, we’ve known of Miocene fossil albanerpetontids for decades, have known of Pliocene specimens since 2005 (Venczel & Gardner 2005), and now know that at least one species persisted into the Pleistocene (Villa et al. 2018). The fact that their fossil record has been creeping towards the Recent means that the possibility of fossil and even extant Holocene specimens being discovered isn’t ridiculous, especially given the small size of these animals and hence tiny size of their bones.
Caption: new salamander species are occasionally discovered in Europe and Asia even now. It would be amazing if an animal suspected to be a ‘new salamander’ one day turned out to be a living albanerpetontid. These reconstructions were published by McGowan & Evans (1995). They might have erred in implying that the scales would be externally visible as shown here; more likely is that they were concealed by epidermis, as in other scaly fossil amphibians. Image: McGowan & Evans (1995).
To be frank, a live albanerpetontid wouldn’t be a particularly spectacular animal: it would be a tiny, slim, salamander-like amphibian less than 10 cm long, and it wouldn’t be much fun to watch since it would spend most of its time hiding and burrowing in leaf litter. But among herp-nerds it would be a huge deal. Live albanerpetontids were scaly-skinned (though the scales were not necessarily visible externally), with eyelids, and with adaptations in the snout, skull-roof, neck and body shape linked to head-first burrowing (McGowan & Evans 1995).
Caption: an artistic reconstruction of a live albanerpetontid… produced for my in-prep The Vertebrate Fossil Record. Image: Darren Naish.
A Eurasian palaeognath. Palaeognaths are the big, flightless ratites (ostriches, emus and so on), the superficially gamebird-like, flight-capable tinamous, and their extinct relatives. A huge amount has been written about the evolutionary history and biogeography of these birds, since their distribution is curious and has resulted in all kinds of different models about how they might have spread around the world. I’ve written about this issue at length on previous occasion (the articles concerned being famous for generating the longest-ever comment threads in the history of TetZoo… though all of this is mostly wasted now, what with SciAm’s paywalling of the site, sigh). Living palaeognaths are absent from Eurasia, despite the former present in the region of ancient, flight-capable Paleogene taxa, extinct ostriches and others.
Caption: as this map shows, modern palaeognaths occurred everywhere until recently (except Antarctica) with the exception of northern North America and the cooler parts of Eurasia. Extinctions across Eurasia, Madagascar and New Zealand of course saw the disappearance of various members of the group. Image: Darren Naish.
The fact that Paleogene Europe was home to many bird groups that no longer occur there but are now denizens of tropical regions elsewhere leads me to hope for a living palaeognath – a tinamou- or bustard-sized species – that descends directly from archaic Paleogene taxa and now lives in the Asian tropics. It should be a cryptic generalist with barred plumage and a mid-length bill and a reduced flight ability.
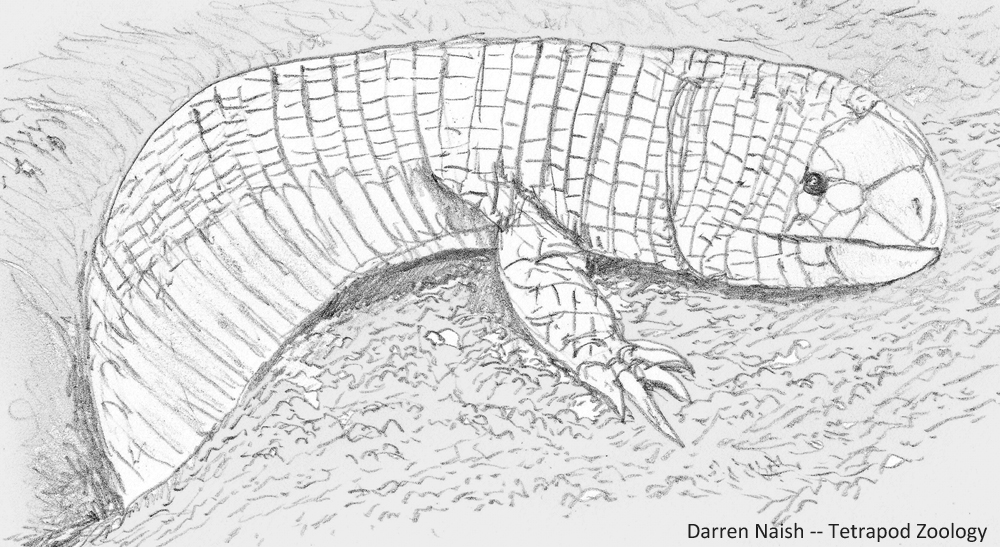
A gigantic, predatory, limbed amphisbaenian. Regular readers of TetZoo might know that I really like amphisbaenians: the mostly limbless, bullet-headed ‘worm lizards’ of the American tropics, Africa, and parts of southern Europe and western Asia. Amphisbaenian evolutionary history and biogeography has become increasingly complex in recent years as we’ve learnt a bunch of new stuff about their fossil history, genetics and anatomy. Among the weirdest of amphisbaenians are the ajolotes (or bipedids), the only extant group to possess limbs. These limbs are not small stumps or flaps (as they are in some other near-limbless, serpentine squamates) but well-developed, clawed forelimbs. According to some phylogenetic models, ajolotes are not the sister-group to limbless amphisbaenians but deeply nested within the limbless clade (Conrad 2008, Videl et al. 2008), in which case their limbedness – if you will – perhaps evolved from limbless ancestors. Add to this the fact that some amphisbaenians are robust-jawed, short-faced predators of vertebrates that ambush prey from beneath the surface and bite chunks from the bodies of surface-dwelling mammals and reptiles.
Caption: Bipes, an ajolote of Mexico (they might occur in parts of the USA as well). Three extant species are recognised. Image: Darren Naish.
So then… where oh where are the giant, limbed, robust-skulled, vertebrate-eating amphisbaenians? By ‘giant’, I am not talking about a graboid-sized monster of several metres (though that would be nice), but a more reasonable animal of a mere 1.5 metres or so. Easily the stuff of nightmares. They could inhabit warm regions of any continent.
Caption: Carl Gans’s illustration of a burrowing ajolote, showing how the large, well-clawed forelimbs function in propulsion. This is clearly a Five-toed worm lizard Bipes biporus; the other extant species have four and three digits, respectively. Image: Gans (1974).
Is there any reason to think that gigantic, predatory, limbed amphisbaenians might actually exist and await discovery? Nope. But I wish it were so. Regular readers might recognise that such creatures are denizens of the alternative-timeline Earth of the Squamozoic, but I’m sure that that’s coincidental.
Caption: what would a gigantic, predatory, limbed amphisbaenian look like? Like this, of course. Image: Darren Naish.
An African or west Eurasian, long-beaked river dolphin. On several occasions within the history of odontocete cetaceans (‘toothed whales’), lineages have moved into brackish and estuarine environments, and eventually made the transition to committed freshwater life. There are the Asian Platanista species, the recently extinct Lipotes of China, and the tropical American Inia species. Once united within Platanistoidea and thought to be close kin, we know today that these animals represent at least three separate transitions to the freshwater environment (the term Platanistoidea is now restricted to the Platanista lineage alone). In addition, members of other groups – I’m thinking of the delphinid Orcaella – occur in rivers within parts of their range. There’s also a fossil beaked whale that might be indicative of freshwater specialisation in yet another odontocete group (Mead 1975).
Caption: river dolphins are pretty special looking. This is a Ganges river dolphin (Platanista gangetica). Image: Zahangir Alom / Marine Mammal Commission / National Oceanic and Atmospheric Administration, public domain (original here).
In view of all this, why aren’t there river-dwelling dolphins in Africa, Europe or western Asia? Again, the answer seems to be… there just aren’t. A few fossil taxa suggest that such animals might have evolved if things had gone another way (there are fossil platanistoids from the Caucasus, for example). But I humbly submit that the great river systems of tropical Africa, the Tigris-Euphrates system of western Asia and the Danube, Po, Ebro, Dniester and others of southern Europe would be much improved if only we knew of their endemic riverine dolphins. I’m talking about a true riverine specialist, convergent with Inia and Platanista, with a long beak, spike-like teeth, reduced eyesight, the works. And if you want to play fast and loose with antiquarian literature and anecdote, there are references in the literature to ‘river dolphins’ in the Nile and there are even one or two eyewitness accounts from central Europe that describe long-beaked ‘dolphins’ seen in rivers and lakes.
Caption: if there are extant west Eurasian or African river dolphins, they should look like this. This is a hypothetical species, modelled on the American Inia and Asian Platanista. Image: Darren Naish.
An endoparasitic tetrapod. Tetrapods have become parasites on several occasions. Vampire bats are parasites of birds and mammals, and it’s even been argued that some blood- and milk-eating human populations can be considered parasites of the mammals they rely on (though the mammals concerned are domesticated, so it’s complicated). Elsewhere among vertebrates, everyone knows about the parasitic catfishes that invade the gills of other actinopterygian fishes and even the urethras of mammals; less familiar is the fact that other actinopterygians can, on rare occasion, become trapped inside the bodies of other vertebrates and then make a successful living. Yes, you read that right. I have in mind the case where two Snubnosed or Pugnose eels Simenchelys parasitica were discovered living inside the heart of a mako shark (Caira et al. 1997; see also Eagderi et al. 2016). This eel is not – despite its name* – ordinarily an internal parasite: this was a case of facultative endoparasitism!
Caption: at left, a snubnosed eel found living inside the heart of a shark. Eels are not tetrapods, it’s true. But here’s evidence that aquatic vertebrates can become endoparasites. Image: Caira et al. (1997). At right: an aquatic typhlonectid caecilian. Surely it’s only a matter of time before we discover an endoparasitic one of those as well. Image: Neil Phillips.
There are all kinds of reasons why a tetrapod couldn’t become an endoparasite, respiration being high on the list. A hypothetical endoparasitic tetrapod would have to be small, with remarkable tolerance of unusual chemical and thermal conditions, with low oxygen requirements, and most likely with the ability to respire cutaneously or via gills. In other words, it should be the world’s weirdest caecilian. As if caecilians aren’t weird enough, I’d love there to be small, endoparasitic caecilians. Given that some caecilians are already aquatic gill-breathers that will consume the tissues of fish (exhibit A: the sequence from River Monsters where Jeremy Wade discovers swarming typhlonectid caecilians in the carcass of a large fish), I predict these animals to be aquatic, South American species that parasitise actinopterygians and aquatic mammals, like Inia the river dolphin.
* Snubnosed eels were given the name ‘parasitica’ because they opportunistically latch on to the bodies of larger fish and eat away at the flesh. They were not thought to ever be proper internal parasites prior to 1992.
And that’s where I’ll stop for now. I actually came up with a list containing numerous additional ‘wish list’ animals but time is against me. Maybe I’ll cover them in another article. Whatever, this was all a bit of fun and I hope you enjoyed it.
For TetZoo articles relevant to the issues covered here, see…
Surreal caecilians part I: tentacles and protrusible eyes, January 2008
Surreal caecilians part II: pass mum’s skin, hold the mayo, January 2008
Close up to Andrias, despite the smell and the teeth, December 2010
Welcome to the Squamozoic!, April 2013
If Bigfoot Were Real, June 2016
PS I’m going to stop linking to the SciAm run of TetZoo articles soon, because I cannot access them at all and they’re now all but useless. They all need to be relocated to an open-access site.
Refs - -
Caira, J. N., Benz, G. W., Borucinska, J. & Kohler, N. E. 1997. Pugnose eels, Simenchelys parasiticus (Synaphobranchidae) from the heart of a shortfin mako, Isurus oxyrinchus (Lamnidae). Environmental Biology of Fishes 49, 139-144.
Conrad, J. 2008. Phylogeny and systematics of Squamata (Reptilia) based on morphology. Bulletin of the American Museum of Natural History 310, 1-182.
Eagderi, S., Christiaens, J., Boone, M., Jacobs, P. & Adriaens, D. 2016 Functional morphology of the feeding apparatus in Simenchelys parasitica (Simenchelyinae: Synaphobranchidae), an alleged parasitic eel. Copeia 104, 421-439.
Graham, G. L. 2002. Bats of the World. St. Martin’s Press, New York.
Mackal, R. P. 1987. A Living Dinosaur? In Search of Mokele-Mbembe. E. J. Brill, Leiden.
McGowan, G. J. & Evans, S. E. 1995. Albanerpetontid amphibians from the Cretaceous of Spain. Nature 373, 143-145.
Mead, J. G. 1975. A fossil beaked whale (Cetacea: Ziphiidae) from the Miocene of Kenya. Journal of Paleontology 49, 745-751.
Naish, D. 2017. Hunting Monsters: Cryptozoology and the Reality Behind the Myths. Arcturus, London.
Simons, E. L. & Ettel, P. C. 1970. Gigantopithecus. Scientific American 222 (1), 77-84.
Venczel, M. & Gardner, J. D. 2005. The geologically youngest albanerpetontid amphibian, from the Lower Pliocene of Hungary. Palaeontology 48, 1273-1300.
Vidal, N., Azvolinsky, A., Cruaud, C. & Hedges, S. B. 2008. Origin of tropical American burrowing reptiles by transatlantic rafting. Biology Letters 4, 115-118.
Villa, A., Blain, H.-A. & Delfino, M. 2018. The Early Pleistocene herpetofauna of Rivoli Veronese (Northern Italy) as evidence for humid and forested glacial phases in the Gelasian of Southern Alps. Palaeogeography, Palaeoclimatology, Palaeoecology 490, 393-403.